Comparator & Reference Listed Drugs (RLD) Procurement
Our Comparator and RLD Procurement Services ensure that your clinical studies have reliable access to the required products, sourced ethically and delivered on time.
Securing high-quality Comparator and Reference Listed Drugs (RLDs) is a critical component of clinical trials. Our Comparator and RLD Procurement Services ensure that your clinical studies have reliable access to the required products, sourced ethically and delivered on time. With our expertise in global sourcing, we mitigate supply chain risks, maintain compliance, and support your research goals with unparalleled efficiency.
-
Global Sourcing Network
Our robust network of trusted suppliers and manufacturers worldwide ensures supply of authentic and high-quality Comparators/ RLDs including:
- Access to hard-to-find drugs
- Verified and licensed suppliers
- Timely and cost-effective procurement
-
Regulatory Compliance
Our procurement processes adhere strictly to international and regional regulatory standards, including:
- Good Distribution Practices (GDP)
- Verification of authenticity and documentation (Certificate of Analysis, Pedigree, etc.)
- Ensuring traceability and audit readiness for all supplies
-
Customized Solutions
- Bulk procurements, Small-batch sourcing
- Assistance in managing expiry and shortages
- Licenses to fulfil Global Tenders
- Compassionate Use, Expanded Access, Managed Access and Programs and Named Patient Programs (NPP)
-
End-to-End Supply Chain Management
From sourcing to delivery, we manage the entire supply chain, ensuring:
- Just-in-time warehousing solutions with temperature-controlled environment and data loggers for distribution
- Shipment Monitoring: Real-time tracking and continuous temperature monitoring and feedback through out the supply chain
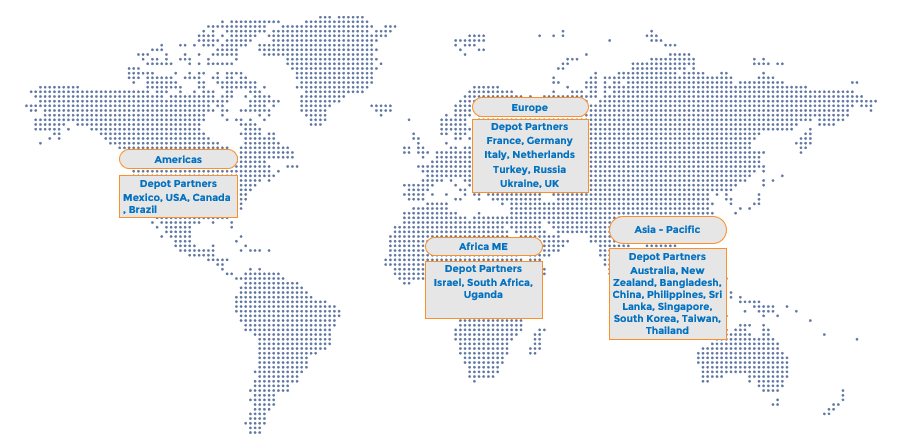
Global Reach, Local Expertise
With operations spanning multiple countries, we combine global capabilities with localized expertise to navigate regulatory and logistical challenges seamlessly.
Why Choose Us?
Our team’s in-depth knowledge and proactive planning ensure uninterrupted supply for your clinical trials, even for high-demand or limited-availability drugs.
We maintain rigorous quality checks at every stage to guarantee the integrity of the products we deliver.
Our strategic partnerships and efficient processes minimize costs, allowing you to allocate resources effectively.
With operations spanning multiple countries, we combine global capabilities with localized expertise to navigate regulatory and logistical challenges seamlessly.
Secure Your Comparator and RLD Needs with Confidence
Partner with us for a streamlined and reliable procurement process.
